Our ERC-funded research evaluates how tissue factor modulates immune signaling by interacting with the interferon-receptor IFNAR1. Tissue factor is the main initiator of the coagulation cascade and is strongly induced during vascular injury and inflammation. We discovered an interaction of TF with IFNAR1: In this heterodimer, both TF and IFNAR1 are inactive. Upon cellular stimulation, this heteromer dissociates, activating simultaneously TF and IFNAR1. Thus, the TF – IFNAR1 heteromer is a singular molecular switch controlling directly thrombo-inflammation. We aim to understand how this heteromer is regulated, on which cells and in which disease conditions the TF – IFNAR1 thrombo-inflammatory switch is relevant, and aim to identify therapeutic approaches to modulate the TF – IFNAR1 heteromer.
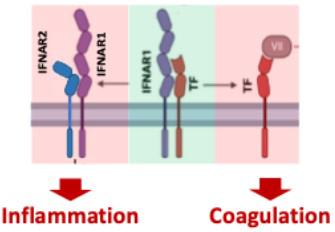 A novel thrombo-inflammatory switch: the TF-IFNAR heterodimer. On resting cells, TF and IFNAR1 form a heterodimer and both are inactive (green). Upon cellular activation, this heterodimer breaks up, IFNAR1 forms a heterodimer with IFNAR2, inducing IFN type 1 signaling upon lingand binding, and TF binds to fVII, initiating blood clotting via the extrinsic coagulation pathway and TF-dependent signaling.
A novel thrombo-inflammatory switch: the TF-IFNAR heterodimer. On resting cells, TF and IFNAR1 form a heterodimer and both are inactive (green). Upon cellular activation, this heterodimer breaks up, IFNAR1 forms a heterodimer with IFNAR2, inducing IFN type 1 signaling upon lingand binding, and TF binds to fVII, initiating blood clotting via the extrinsic coagulation pathway and TF-dependent signaling.